|
||||||||||
Date: May 3, 2024
by Chaya Venkat
Related Articles:
GVHD

Transplant statistics are reported on groups of patients that span the whole gamut. Most often they are not even limited to CLL, but include patients with much more aggressive and dangerous blood cancers. But even more important, the patient cohorts are very diverse in their health status going into the transplant. Some can run marathons and some are basket cases from the get go. So, how is one to make a half-way decent guess at one’s chances of sailing through the transplant procedure and come out with a bona fide CLL cure at the other end? The overall statistics reported may be scaring away patients who might actually have much better chances of success than they think.
Here is a cheat-sheet of what it takes to ace the transplant process:
We have written before about GVHD (Graft versus Host Disease). I will not go into the details here, just the broad brush strokes of why GVHD happens.
Basically, patients go into transplants right after pre-conditioning (usually with our ever popular fludarabine, as well as low dose radiation). Radiation and chemotherapy target rapidly dividing cells, since this is the common profile of cancer cells. However, another set of cells that divide rapidly as normal part of their functioning are skin cells and mucosal cells lining your mouth, stomach and gut. These cells are sloughed off daily as part of normal wear and tear, and must replenish their numbers to keep up with the losses. It is therefore no surprise that your skin and mucosal linings of the mouth, stomach, gut etc often take a beating during chemo and radiation therapy, leading to skin and gut inflammation.
Now the new graft comes in, with its bright eyed and bushy tailed new immune system cells ready and anxious to go to work. All that eager enthusiasm of the new recruits means that when they spot the inflamed cells of the skin, gut etc, they assume the worst, they assume an ongoing infection is causing the inflammation. Donor T-cells and other immune system cells from the graft flood into the area, and we can only describe the mayhem that ensues as a case of friendly fire gone berserk. In a nutshell, GVHD happens because the new graft cannot yet tell the difference between friend and foe, and the over-kill makes the initial chemo induced inflammation far worse.
Our skin and lining of the gut are important barriers between the nasty bugs and stuff out there in the world and our vulnerable insides. Inflammation often causes breakdown in this important barrier function, which in turn can lead to dangerous infections. You can see why it is important to bring this inflammation under control, and quickly. The standard approach to controlling GVHD is to use immune suppressants (steroids like prednisone, or other immune suppressive drugs such as cyclosporin). Putting the brakes on the functioning of the newly grafted donor T-cells and other effector cells often accomplishes what is required, and GVHD is generally brought under control by this approach.
What is wrong with using massive doses of immune suppressing drugs to control GVHD? Well, while the new immune system is taking a little nap, weaned away from doing GVHD damage and lulled into complacency by the immune suppressive drugs, it is not doing much of anything else either. Like not keeping an eye out for legitimate infections; like not going after the remaining traces of the CLL cells still lurking around; like not doing its job in all the ways an immune system is supposed to do. You get the picture. Put the brakes on too hard to control GVHD, you can also say bye-bye to effective GVL, and effective protection from infections. Patients who develop heavy duty GVHD and therefore need aggressive immune suppression for long periods of time to control it, are also at increased risk of life threatening infections, as well as relapse of the CLL because the new immune system does not go out and hunt down the last of the CLL cells, before the cancer has a chance to re-establish itself.
It has been observed that patients whose GVHD took the form of skin rash did a lot better than patients who had extensive mucosal damage to their GI tract. The explanation is that skin problems are often dealt with topical steroidal creams or other drugs that work locally. The drugs do what is needed, control the skin inflammation, but that is the extent of their impact. They do not go where they are not wanted. Limiting systemic availability of the immune suppressive drug means the patient can still get benefit of GVL and infection fighting efforts of the newly grafted immune system cells, even though skin GVHD is under control.
Unfortunately, it is not possible to treat GVHD of the gastrointestinal (GI) tract with the usual topical drugs, and as many as 60% of transplant patients have to deal with this nasty form of GVHD. When drugs such as prednisone are given as oral tablets, the steroid is absorbed in your GI tract and gets everywhere in your body. The immune suppressive effects of the steroid are felt through out the body, and it is no surprise that along with suppression of the unwanted GVHD, we also have suppression of the ever-so-necessary GVL and infection fighting capability. Long term systemic use of steroids also carries well known penalties. What we need is a drug that is potent where it needs to be, but does not mess up or hobble the immune system too much in the rest of the body.
A number of new approaches are being tried to control GVHD without shutting down much needed GVL. One idea is to control the initial inflammation that attracts the attention of the new graft, causing all those friendly-fire accidents. Believe it or not, a little yogurt to settle the stomach is not a bad idea, it seems! A recent editorial in Blood was aptly titled “Let them eat Yogurt”. Soothing the lining of the gut and stomach with pro-biotics found in yogurt is good for reducing chemo and radiation induced inflammation, and this helps reduce subsequent GVHD. Let us know if you want to read the full text of this editorial, as well as the article it discusses.
Blood. 2024 Jun 1;103(11):4365-7. Epub 2024 Feb 12
Probiotic effects on experimental graft-versus-host disease: let them eat yogurt
Gerbitz A, Schultz M, Wilke A, Linde HJ, Scholmerich J, Andreesen R, Holler E.
Department of Hematology/Oncology, Institute of Medical Microbiology and Hygiene, University of Regensburg, Germany.
Acute graft-versus-host disease (aGVHD) often limits feasibility and outcome of allogeneic bone marrow transplantation. Current pathophysiologic concepts of aGVHD involve conditioning regimens, donor-derived T cells, proinflammatory cytokines, and bacterial lipopolysaccharide (LPS) as a major trigger for aGVHD. LPS derives mostly from gram-negative bacteria and can enter circulation through the impaired mucosal barrier after the conditioning regimen. Probiotic microorganisms have been shown to alter the composition of the intestinal microflora and thereby mediate anti-inflammatory effects. We hypothesized that modifying the enteric flora using the probiotic microorganism Lactobacillus rhamnosus GG, would ameliorate aGVHD. Here we show that oral administration of Lactobacillus rhamnosus GG before and after transplantation results in improved survival and reduced aGVHD. Furthermore, subculturing of mesenteric lymph node tissue revealed a reduced translocation of enteric bacteria. Our findings suggest that alteration of the intestinal microflora plays an important role in the initiation of experimental aGVHD.
PMID: 14962899
____________
Another approach being tried is to separate out the donor T-cells in the graft coming in, hold back from giving them to the transplant patient for a while. The idea is to withhold these potent immune system cells until things settle down a bit. Once the rest of the graft is nicely settled down, the patient has recovered from the effects of the pre-conditioning chemo and radiation, inflammation is under control etc, then the donor T-cells are given back to the patient. This second bit is important, giving the donor T-cells to the patient at the appropriate time. T-cells are very important in protecting us from infections, and they also play a major role in hunting down the remaining traces of cancer cells in the body. Patients who did not get back the donor T-cells at some point had far more infections, and more cases of cancer relapse.
Variations on the theme of this approach are being studied at a number of research centers. I am most impressed with the clinical trial at the National Institute of Health (Bethesda, MD) that is doing some of the cutting edge research in this area. We reviewed their clinical trial in a recent article Stem Cell Transplants Almost Free. If you are a mini-allo transplant candidate, and you have a matched sibling, this trial is very worth exploring. Let us know if you are interested in this program and want us to help you contact the researchers at the NIH.
The two items above are by way of appetizers leading up to the main entrée for this article. A new and improved drug to control GVHD is on the horizon. For a change, we don’t have to wait years and years for it to become available - this drug is going up for FDA approval this summer, and I am willing to bet dollars for donuts that it will get approved. A large scale, multi-center and double arm placebo controlled clinical trial of this new drug has demonstrated benefits that knocked my socks off! It does a great job of controlling GVHD, and seems to do that without tying the hands of the new graft when it comes to controlling infections or going after remaining cancer cells. Credible research, credible institutions doing it, a robust clinical trial design that allows us to have confidence in the statistics reported – I like it, and I wish we had equally reliable results in other areas.
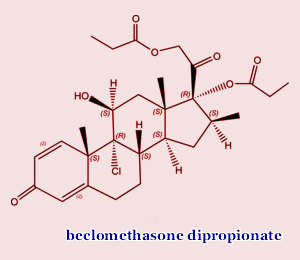
Beclomethasone dipropionate may well be the drug that significantly improves the survival statistics for transplants. Don’t you love these long winded chemical names for drugs? Fortunately for you, we are going to use the three letter acronym BDP instead. Below is an abstract of a very recent article in the prestigious journal Blood. If you are contemplating a transplant now or some time in the future, the full text of this article is mandatory reading, in my opinion. Do write to us if you want help in locating the full text PDF.
Blood. 2024 Jan 23; [Epub ahead of print]
A randomized, placebo-controlled trial of oral beclomethasone dipropionate as a prednisone-sparing therapy for gastrointestinal graft-versus-host disease
Hockenbery DM, Cruickshank S, Rodell TC, Gooley T, Schuening FG, Rowley SD, David D, Brunvand M, Berryman B, Abhyankar S, Bouvier ME, McDonald GB.
Fred Hutchinson Cancer Research Center & University of Washington School of Medicine, Seattle, WA.
We tested the hypothesis that oral beclomethasone dipropionate (BDP) would control gastrointestinal graft-vs-host-disease (anorexia, vomiting, and diarrhea). Patients were randomized to prednisone for ten days and either oral BDP 8 mg/day (N=62) or placebo (N=67) tablets for fifty days. At Study Day-10, prednisone was rapidly tapered while continuing study drug. On an intent-to-treat basis, the risk of GVHD-treatment failure was reduced for the BDP group at Study Day-50 (hazard ratio 0.63, 95% CI 0.35-1.13) and at 30 days follow-up (HR 0.55, 95% CI 0.32, 0.93). Among patients eligible for prednisone taper at Study Day-10, the risk of GVHD-treatment failure was significantly reduced at both Study Days-50 and -80 (HR 0.39 and 0.38, respectively). By day-200 post-transplant, 5 patients randomized to BDP had died, compared to 16 deaths on placebo, a 67% reduction in the hazard of mortality (HR 0.33, p=0.03). In 47 recipients of unrelated and HLA-mismatched stem cells, mortality at transplant day-200 was reduced by 91% in the BDP group, compared to placebo (HR 0.09, p=0.02). The survival benefit was durable to one-year post-randomization. Oral BDP prevents relapses of gastrointestinal GVHD following tapering of prednisone; survival is statistically significantly better among patients receiving BDP.
PMID: 17244684
_____________
Because GVHD typically involves the lining (“mucosa”) of the gut all the way from one end to the other (your stomach to your rectum), the researchers formulated BDP into a set of two tablets. One tablet dissolves immediately upon coming into contact with the acid in the stomach. This makes the drug available to the lining of the stomach and the jejunum (the part of the GI tract that comes immediately after the stomach). The second tablet is enteric coated, and does not dissolve in the stomach but further down the line, making the drug available to the later part of the GI tract, including the small and large intestines.
BDP has strong immune suppressive activity. Furthermore, BDP is metabolized by the GI tract mucosa and liver, converted to another compound called 17-BMP, which is roughly 25 times more active than even BDP. High concentrations of BDP and 17-BMP at the mucosal lining of the GI tract lead to effective control of GVHD inflammation. The parent drug BDP cannot be detected in the rest of the body, and has no impact on systemic immune suppression. Its offspring 17-BMP is detected in general circulation. However, compared to prolonged use of prednisone to control GVHD, any systemic effect of 17- BMP are thought to be relatively minor, since patients randomized to BDP had infrequent fatal infections, fewer cancer relapses and better day-200 post-transplant survival.
This was a Phase-3, randomized and placebo controlled clinical trial. All participants were stem cell transplant patients. All received 10 days of prednisone, to bring GI tract GVHD under reasonable level of control. This is pretty much standard procedure for transplant patients, and only those that achieved GVHD control at this point were allowed to continue with the trial. After the 10 days of prednisone, 62 of the participants went on to get 8 mg/day of oral BDP tablets (pair of tablets, four times each day) for 50 days. The other 67 participants got a placebo.
Now we get to the juicy part, the results. Here is how things stacked up, 200 days later. I don’t know about you, but I was pretty impressed by these results. Use of BDP knocked percent of patients who had died by day 200 from 24% to 8%. This improvement came about as a result of improvement in reduced fatal infections, as well as reduced cancer relapse. We seem to have achieved success in controlling GVHD, without losing the all important graft-versus-leukemia, or protection against infections. These are statistically significant results folks.
| Outcome | Placebo | BDP |
| Number of Patients | 67 | 62 |
| % of Patients Who Died | 24% | 8% |
| Due to Infection | 13% | 5% |
| Cancer Relapse | 13% | 5% |
| Some patients had both infection and relapse of their cancers as cause of death | ||
It is well known that GVHD is often far worse in patients who are not lucky enough to have perfect 10 point matched donors. (You can read more about stem cell donor matching in our article Matching Made Simple). So, how did BDP work for patients who were particularly at risk of high grade GVHD, because they had partially mis-matched donors? There were 47 patients with this profile in this study. For this high risk group, the impact of BDP was nothing short of stunning.For these guys, there was a 91% reduction in patient mortality at the 200 day point. Wow. Now that is something to write home about, wouldn’t you say?
BDP is not a new drug, and the clinical trials testing its effectiveness in post-transplant GVHD go back all the way to 1995. I have tracked publications by the lead author Dr. G. B. McDonald on this subject for this length of time. The concept of using a steroid drug that has limited bioavailability, either because of the way it is metabolized or because of enteric coating formulation, that is not a new concept either. “Budesonide” has become the gold standard steroid therapy for Crohn’s disease. Crohn’s disease is chronic and potentially very debilitating autoimmune disease of the GI tract, not unlike the GVHD we are discussing here. Patients often need to be on steroids for long periods of time, and classic drugs such as prednisone carried the risk of systemic absorption of the drug causing the usual adverse effects associated with long term use of steroids. Budesonide became the gold standard specifically because its action is limited to the GI tract almost exclusively.
See if you can wrap your brain around this scenario: BDP that has been known at least since 1995, and the concept of targeted delivery of drugs to just the GI tract has also been known for quite a while. So, how is it that we are just getting around to reading about late stage trials and possible FDA approval for BDP later this summer? Welcome to the real world of medical research, my friends. This is how long it takes for drugs to wind their way through Phase I, Phase II, and the pivotal multi-center, double arm large scale Phase III trials such as this. This latest Phase III trial took three years to recruit patients, from July 2024 to July 2024. And it took from July 2024 to January 2024 for the fully analyzed results to be published.
That is roughly 6 years from start of this important late stage trial to final publication of results. I will leave it up to you to judge how long it will take the information to percolate slowly through the process of CME (continuing medical education) to all the various transplant centers, how long before turf issues and not-invented-here ego problems make way to acceptance of the new approach to controlling potentially life threatening GI tract GVHD in transplant patients. There is not much we can do about the 6 years it took for this clinical trial. But perhaps we as informed and interested patients can do something about getting the message out to our healthcare providers without undue delay. If you are a transplant candidate and you read this review, and it tempts you enough to ask to read the full text of the article, I am willing to bet you will ask your transplant team whether they plan to use BDP to take care of your GVHD down the road.
As I said above, I expect this drug will be in front of the FDA panel for review and approval sometime this year. Will it get FDA approval? I think so. The results are pretty substantial, and the work was done at a number of very prestigious institutions. This latter was an important bit of information for me as I evaluated this report, since there was an obvious conflict of interest. Dr. McDonald, the principal investigator in much of the BDP research, is also the founder of and retains an ownership interest in, DOR Biopharma (www.dorbiopharma.com), the company that makes and will market the oral version of BDP.
Almost every day I get emails from patients asking how long it will be before they can get access to some new drug that showed excellent results in mice, or cell lines in a test tube. The sad fact of life is that many of these “miracles of the month” will fail in subsequent pre-clinical testing, and many more will fail to show sufficient efficacy without toxicity in early phase trials. Only a handful will find their way into well designed large-scale multi-center trials such as this one. Even for the success stories, we are looking at very long lead times needed to translate the lab discovery to the patient bedside. What is the solution to fix this problem? I am not sure I know — and I doubt if there is anything anyone can do to speed things up, not without substantially short-circuiting safety issues. But this much I can tell you. Keep a large pinch of salt handy when you read the next gushy PR-style report, complete with touching human interest stories, suggesting the easy fix cure for CLL is right round the corner. For now, for most of us with CLL right this minute, a mini-allo stem cell transplant is still the only real cure out there.
 Enter Keywords: |
———
Disclaimer: The content of this website is intended for information only and is NOT meant to be medical advice. Please be sure to consult and follow the advice of your doctors on all medical matters.
Copyright Notice:
Copyright © 2024-2007 CLL Topics, Inc. All Rights Reserved.
All materials contained on this site are protected by United States copyright law and may not be reproduced, distributed, transmitted, displayed, published or broadcast without the prior written permission of CLL Topics, Inc. You may not alter or remove any trademark, copyright or other notice from copies of the content.
However, you may download and print material from CLLTopics.org exclusively for your personal, noncommercial use.
———