|
||||||||||
Date: January 3, 2026
by Chaya Venkat
Index Pages of Related Articles:
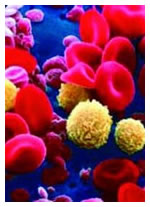
Stem cell transplants are intimidating. They are no springtime walk in the park. It is hard to figure out who should go this route — and when to do it. And they are not cheap. Depending upon the center doing the transplant and the complexities of the specific case, a stem cell transplant can cost as much as a quarter of a million dollars. Many insurance companies explicitly rule out stem cell transplant coverage. You were out of luck on this important procedure if you were uninsured (or under-insured!), not independently wealthy, or simply happened to be in a region where stem cell transplants are not easily available (which is just about everywhere outside the USA, Western Europe and a few other countries)
Not any more. Consider this our New Year present to the CLL community. Here is our review of an opportunity for CLL patients to get state-of-the-art care stem cell transplants at the National Institutes of Health, (National Heart, Lung and Blood Institute, NHLBI) Bethesda, Maryland, USA — almost entirely free of charge. And you do not even have to be an American to get it! This information is time-sensitive folks. I doubt the offer will be open for very long. Read on, and act promptly if this wonderful gift is of interest to you.
Bear with me while I highlight the importance of stem cell transplants for certain CLL patients. It is kind of important that you understand the when, why, how and who of this complicated business.
CLL comes in many flavors, and it is very important to know what kind you have. Just a few years ago, researchers running clinical trials reported overall response rates and remission times obtained using a mixed group of patients. No one could quite predict why some CLL patients responded well to just about any drug and why some unlucky ones did not respond worth a damn to any chemotherapy drug or combination of drugs. Much progress has been made on this front, with the advent of modern prognostic indicators. (Some background reading if you are new to all or any of this: What Type of CLL Do You Have?, Prognosis at Diagnosis, Dawn of a New Era).
Of particular interest is the FISH test (Fluorosence In-Situ Hybridization), which looks for presence of the common chromosomal aberrations found in CLL cells (FISH-ing for Answers). A 17p (P53 gene) deletion is the most serious defect of the lot and patients with this particular deletion reported in their FISH test are very likely to have an aggressive form of the disease. These patients are likely to need therapy sooner, less likely to get a good response and get shorter remissions after therapy. Single agent fludarabine (the so-called “gold standard”) does not work in 17p deleted cases. Neither does Rituxan. Byrd, et al. published a seminal paper a couple of years ago, showing that patients with this deletion do not respond well to the RF (Rituxan + fludarabine) combo therapy either (RF Therapy: Not All Patients Have the Same Risks and Benefits). We hope more and more researchers follow this trend and break down their response statistics by prognostic markers. Bottom line: people with 17p deletion have few and not very good options today.
The major topic of conversation and much of the research presented at the latest ASH conference (ASH 2026 Highlights) was about treating CLL patients with poor prognostics. The holy grail of CLL research now is developing successful therapy regimens for patients with the dreaded 17p deletion. This is important for all of us chickens, not just the folks who have this adverse prognostic indicator right now. How so, you ask. Well, there is something called clonal evolution in CLL. You may start out with a very benign and good prognosis 13q deletion, but there is a reasonable chance that your CLL will gradually acquire other deletions as time goes by, especially if you used aggressive chemotherapy regimens, and even more especially if you have unmutated IgVH gene status. CLL cells, especially the IgVH unmutated variety, have unstable genomes that can get bent out of shape and, break off pieces of their chromosomes easily, especially when they are roughly handled and mangled by chemotherapy. (You can read more in our article titled Clonal Evolution.) Roughly half of the patients who had relapsed after fludarabine therapy had the 17p deletion, even though far fewer had this cross to bear before going into therapy. You get my drift? A large percent of us will have to walk this path, at some point in our CLL journey. That is why I applaud all the research going into treating 17p deleted CLL patients.
The only therapy regimen that has proven to fully CURE a significant percentage of patients is a stem cell transplant (The Only Real Cure). The good news is that allogeneic (donor) stem cell transplants don’t seem to care about the FISH status of patients, provided the patient goes into the transplant with only minimal residual disease. “Mini-allo" transplants have come a long way and the survival statistics have improved a great deal after switching to this “Reduced Intensity Conditioning” or “RIC” approach in getting patients ready prior to receiving the precious graft of stem cells from a generous donor. (The term mini-allo is sort of slang for RIC, also called non-myeloablative stem cell transplants). The other major advantage of mini-allo transplants is that it is possible to use this “gentler” approach with older patients. Typically, full strength myeloablative transplants are not done in patients over 55 years. Bit of a bummer, considering that most CLL patients are in this group: the median age at diagnosis of CLL is over 55! Fortunately for our senior citizens, mini-allo transplants are easier on the body and therefore do not have a similar age limit. I have recently heard of a 70-year old who had the procedure.
Should you wait out until the last bitter moment before you opt for a transplant? Is a stem cell transplant a last ditch effort, only to be undertaken when you have flunked all other options? I do not think so. And that opinion is endorsed by many CLL experts. If you have aggressive CLL and poor prognostic indicators that suggest you may not respond very well to conventional therapy, it is important to realize that a stem cell transplant is in your future, like it or not. As the abstract below points out, patients who head for the transplant center while they are still in good shape do a lot better than those that have waited too long. Too many unsuccessful chemotherapy regimens under your belt make you a poorer candidate for a stem cell transplant. If you are relatively young with family to raise, poor prognostics, not likely to run the clock out on the CLL by sequential low intensity therapies to keep it under control, you may want to discuss with your doctors the timing of a stem cell transplant.
Biol Blood Marrow Transplant. 2026 Oct;12(10):1056-64
Predictors of improved progression-free survival after nonmyeloablative allgeneic stem cell transplantation for advanced chronic lymphocytic leukemia.
Brown JR, Kim HT, Li S, Stephans K, Fisher DC, Cutler C, Ho V, Lee SJ, Milford EL, Ritz J, Antin JH, Soiffer RJ, Gribben JG, Alyea EP.
Department of Medical Oncology, Dana-Farber Cancer Institute, Boston, MA 02115, USA.
Although chronic lymphocytic leukemia (CLL) remains an incurable disease with standard chemotherapy, the appropriate role and timing of transplantation are unclear. In this analysis, we report the outcomes of 46 patients with advanced CLL who underwent nonmyeloablative stem cell transplantation (NST) from HLA-matched unrelated (67%) or related (33%) donors. Fludarabine (30 mg/m2 x 4) and low-dose intravenous busulfan (0.8 mg/kg/day x 4) were used for conditioning. The 2-year overall survival (OS) and progression-free survival (PFS) rates in this refractory patient population were 54% and 34%, respectively, with a median follow-up of 20 months. The primary cause of treatment failure was relapse, with a 2-year cumulative incidence of 48%. High hematopoietic donor chimerism > or = 75% at day +30 was a significant predictor of 2-year PFS (47% vs 11%; P = .03). In multivariate analysis, chemotherapy-refractory disease at transplantation was associated with a 3.2-fold risk of progression (P = .01) and a 4.6-fold risk of death (P = .02). Increasing number of previous therapies and increasing bone marrow involvement were also associated with decreased PFS and OS. These results suggest that NST using fludarabine and low-dose intravenous busulfan is a reasonable treatment option for patients with advanced CLL, but that NST earlier in the disease course will likely be needed to achieve long-term disease control in a high proportion of patients.
PMID: 17084369
____________
The authors are quite clear and blunt in their message: patients who have had more chemotherapy regimens under their belt and who have an increased amount of CLL in their bone marrow are also more likely to relapse after the transplant — and more likely to die. Patients who go in too late, whose CLL is refractory to chemotherapy have much poorer chance of success with the transplant. These guys are more than three times as likely to have the CLL come back after the transplant and just under five times as likely to die!! Not the kind of odds I like to have.
The moral of the story: if you have poor prognosis CLL, you had better learn to be bloody minded in evaluating your options. Dithering until the last moment looking for miraculously curative approaches (not!) that waste precious time is not such a good idea. The remission you get after your very first therapy is likely to be the best one you will get. As one of the CLL experts put it, it is not a good idea to waste the window of opportunity. These patients who will ultimately need a transplant are better off if they face up to it sooner rather than later.
Getting yourself organized for a stem cell transplant is no easy task. First, there is the question of the donor. In the majority of cases, the donor is a brother or sister: and that means getting yourself and your siblings tested for compatibility. If none of your siblings is a match, and only then, a search is conducted for matched unrelated donors (MUD) in the stem cell banks. The process can take anywhere from a couple of weeks (for sibling matches) to several months for MUD matches. Better get your ducks in a row early on, even if you are not going to use this transplant card right away!
Where to get the transplant is a really important issue. Transplants are a team effort, with many experts with different areas of expertise participating in the complex procedure. We are still in the steep part of the learning curve. Centers that do a lot of transplants, that have well-honed procedures and transplant teams that work well together, have better success statistics to show for it. You may be entirely happy with your local oncologist or even your nearby CLL expert. But that does not automatically mean you should get your transplant done locally. This is one of the biggest decisions you will ever make — it is literally a life or death decision. Make sure you are smart about choosing your transplant center. Expertise is the name of the game, not commuting convenience.
Money and logistics are always important. Does your insurance cover stem cell transplants? Better check ahead of time! Almost always, insurance will not cover incidental costs, such as travel to and from the transplant center. It is often required that you stay locally for several months (2-3 months) immediately after the transplant, so that your progress can be closely monitored. It is also very important that you have a devoted caregiver stay with you during this period, to take care of you and the million little details that have to be handled.

I was fortunate to get an introduction to Dr. John Barrett, Chief of the stem cell transplantation section of the National Institute of Health (National Heart, Lung and Blood Institute). Here is his professional profile Dr. John Barrett, NIH curriculum vitae, publications list, picture and all. You can read all about the clinical trial of interest to us here: http://clinicaltrials.gov/show/NCT00079391. Below is a shorter description, as well as contact information, followed by my cheat-sheet summary.
Fact Sheet:
Open treatment protocols for Patients with Hematologic Malignancies at the National Heart, Lung and Blood Institute
National Institutes of Health, Bethesda MD
Allogeneic HLA-matched sibling stem cell transplant protocols:
At NHLBI we have stem cell transplant protocols for any patient aged 10-75 years with an HLA identical sibling in whom allogeneic stem cell transplantation is the standard of care.There is no charge for patients or donors on NIH study protocols for clinical care and follow-up, but travel for the first visit is not compensated.
Screening: Prior to referral we can help with screening patients for eligibility and perform HLA typing. Before signing consent, suitable transplant candidates and their donor undergo a final eligibility screening at NIH.
Transplant: According to age, patients receive a standard or reduced intensity T cell depleted or T cell modulated transplant aimed at preventing graft-versus-host disease.
Follow-up: For the first 3-4 months post transplant the patient and a care giver remain in the Bethesda area for close monitoring; thereafter they return to their referring physician for regular follow-up. Long-term follow-up at NIH is peformed at 6-9 months post-transplant and yearly thereafter.
Program Director: John Barrett MD, Chief, Stem Cell Allotransplantation Section, NHLBI, NIH
Tel 301-402-4170, Fax 301-480-2664
Contacts:
| Research Nurse Coordinator | Principal Investigator |
|---|---|
| Laura Musse, RN, CRNP | Stephan Mielke, M.D |
| Tel 301-496-3841, FAX 301-480-0758 | Tel 301-402-5294, Fax 301-480-2664 |
 |
 |
Biol Blood Marrow Transplant. 2026 Dec;12(12):1318-25
T cell depleted peripheral blood stem cell allotransplantation with T cell add back for patients with hematological malignancies: effect of chronic GVHD on outcome.
Montero A, Savani BN, Shenoy A, Read EJ, Carter CS, Leitman SF, Mielke S, Rezvani K, Childs R, Barrett AJ.
Stem Cell Allotransplantation Section, Hematology Branch, National Heart, Lung and Blood Institute, Bethesda, Maryland 20262-1202
One hundred thirty-eight patients with hematologic malignancies received myeloablative T cell-depleted peripheral blood stem cell transplant (PBSCT) from an HLA-identical sibling donor. The T cell dose was adjusted to 0.2-1 x 10(5) CD3(+) cells/kg. The CD34 dose was 2.7-16 x 10(6)/kg. Patients with acute graft-versus-host disease (GVHD) grade <2 received 1 or 2 donor lymphocyte infusions of 10(7) CD3(+) cells/kg between days 45 and 100. Patients were designated according to relapse probability as standard or high relapse risk (77 and 61, respectively). Overall survival (OS), relapse-free survival, relapse, and transplant-related mortality (TRM) were 58%, 46%, 40%, and 20%, respectively, after a median follow-up of 4 years. Fifty-three (39%) and 21 (15%) patients developed grade 2-4 and 3-4 acute GVHD. Forty-two (36%) had limited and 29 (25%) had extensive chronic GVHD. In multivariate analysis, disease risk was an independent factor for OS and relapse, day-30 lymphocyte count for OS and TRM, and chronic GVHD for OS and relapse. PBSCT with early T cell add back leads to comparable rates of chronic GVHD compared with T cell-replete PBSCT. However, this chronic GVHD after T cell add back is associated with less mortality and retains a protective effect in terms of relapse, at least in the standard-risk patients.
PMID: 17162214 [PubMed - in process]
____________
If you are one of our members for whom this option makes sense, I urge you to contact the people running this clinical trial and make sure you fit their inclusion criteria, right away. The NIH is a good place to get your transplant, especially if it is free of charge! Do write and let us know if you are going to be a participant in the trial, since we like to keep track of our members and how they are doing on the clinical trials we recommend. And if there is any thing we can do to help expedite the process of your participation, if you have questions we can answer, don’t hesitate to write as well. Good luck!
 Enter Keywords: |
———
Disclaimer: The content of this website is intended for information only and is NOT meant to be medical advice. Please be sure to consult and follow the advice of your doctors on all medical matters.
Copyright Notice:
Copyright © 2026-2007 CLL Topics, Inc. All Rights Reserved.
All materials contained on this site are protected by United States copyright law and may not be reproduced, distributed, transmitted, displayed, published or broadcast without the prior written permission of CLL Topics, Inc. You may not alter or remove any trademark, copyright or other notice from copies of the content.
However, you may download and print material from CLLTopics.org exclusively for your personal, noncommercial use.
———